How Animals Code Their Kids for Survival
Insights into epigenetics and inheritance show that some organisms can adapt to a changing world.
Article body copy
The week before my son flew across the country to start university in Nova Scotia, I found myself spewing advice at breakneck speed. A quick stop at the ATM triggered sage wisdom about investing for the future and how to avoid credit card debt. The grocery store elicited reminders about buying in bulk and the current state of play around sell-by dates. “Be gentle with yourself. Keep in touch with your sister. Brush your teeth!” Into one long weekend of errands and packing, I condensed 18 years of parenting dogma.
My actions seemed odd—even to me. And yet I felt compelled. For what else is parenting, if not an insatiable drive to provide one’s offspring with the best chance to flourish in an unknown future? The information passed from one generation to the next, as biographers ceaselessly demonstrate, can be the making or the ruin of us.
So how, I wondered, do other species prepare their offspring for uncertain futures? Sure, all animals and plants inherit characteristics that evolve through changes in genes over generations. But I wanted to know more about immediate time. Are there things parents of other species do to try to improve their kids’ chances for survival in this rapidly changing world?
My hunt for information eventually leads me to a most unexpected source—a May 1, 2018 media release from the ARC Centre of Excellence for Coral Reef Studies in Australia that reads: “Reef fish inherit tolerance to warming oceans,” with the intriguing subtitle, “Thanks to mom and dad, baby reef fish may have what it takes to adjust to hotter oceans.”
The idea is so captivating I find myself flipping between the online recipe for quinoa burgers I am preparing and YouTube videos of lead researcher Philip Munday explaining how baby fish benefit from their parents’ experiences. My keyboard becomes so sticky from all the times I have to stop cooking to type a note that I give up and plop down in a chair to give Munday my full attention. This fascinating research is changing my brain. When I listen to him, the idea of animals as helpless victims trapped by their instincts to face an inevitable future of destruction slips away. Baby fish are shaped by the experiences of their parents. Fish are resilient.
“What we are finding,” he says when I call him a few days later on Skype, “is a surprising amount of potential for fish to cope.”
He explains by telling me about spiny chromis damselfish, which he and his team raise in the lab. Most coral reef fish are negatively affected by a water temperature increase of just a degree or two above the summer average. The change can impact swimming ability or growth rate and, since warm water holds less oxygen, their capacity to breathe also suffers.

Spiny chromis damselfish (Acanthochromis polyacanthus) raised in water a few degrees warmer than normal have offspring that are better able to survive increased temperatures. Photo by Colin Marshall/Minden Pictures
Yet remarkably, when Munday’s lab reared spiny chromis damselfish, and then their offspring, in water 3 °C warmer than current-day ocean temperatures, the offspring had a perfectly normal capacity to get enough oxygen. The baby coral reef fish were able to handle higher water temperatures when their parents were previously exposed to those conditions.
It turns out the capacity for offspring to benefit from their parents’ experiences doesn’t just happen with fish. Munday tells me about Daphnia, often called water fleas, that are found in freshwater lakes, ponds, and puddles. The tiny crustacean can hatch with either a round head or a pointed head. If it shares the water with predators such as fish or midges or other insects, spikes and spines help lessen the likelihood of being eaten. For many species of juvenile water fleas, whether or not they grow a defensive helmet depends on the experiences of their mother. “If a mother Daphnia has been exposed to chemical cues of a particular predator while she is pregnant,” Munday explains, “then she produces lots more offspring with pointy heads.”
Such astonishing malleability exists in other species too. “If aphids are in a habitat with predators, their babies can develop wings and fly away. They have the plasticity to have wings or not,” says Munday.
It sounds like a fairy story: the environment changes and the hero grows wings! But the dark force driving Munday’s research is all too real. What he wants to know is how animals are affected by climate change, and if they’re able to adapt.
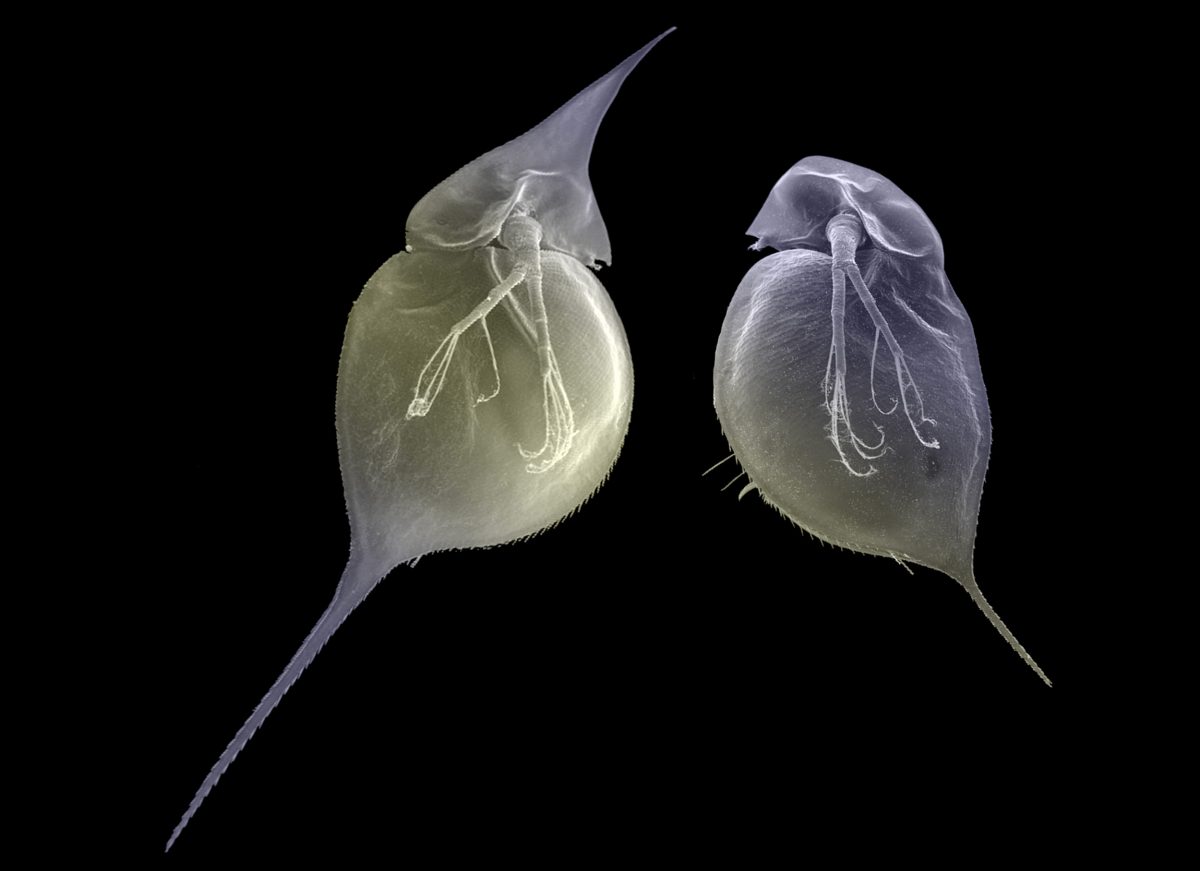
Water fleas, such as the Daphnia lumholtzi shown here, can be born with or without spikes or spines. Whether they grow these adornments depends on the experiences of their mother. If she’s exposed to predators in the water, her offspring are born with a defensive “helmet.” Photo by Christian Laforsch/Science Photo Library
Changes in climate are already exposing fish to warmer ocean temperatures and ocean acidification. Munday’s work fuels a recent surge of interest that seeks to understand the capacity of offspring to benefit from their parents’ experiences in these rapidly changing times. Part of the answer, Munday says, lies within the science of epigenetics.
Epigenetics is the study of changes in gene expression. It differs from a mutation in that the actual sequence of a gene—the underlying DNA code—remains unchanged. Think back to those damselfish. Placing the parents in warmer water doesn’t change the basic genetic code they pass on to their babies. But it can influence which genes are expressed. It’s the exposure of the parents to the higher water temperatures that appears to determine which genes in the DNA sequence are turned on or off. The parents already had to have the capacity to deal with warmer temperatures somewhere within their existing DNA in order for it to be able to be turned on, or expressed, within their babies.
The potential for differences in gene expression depends upon the plasticity of forms that already exist within embryos in a wild population. And that’s what makes coral reef fish such great study subjects for epigenetics—they embody some of the world’s most amazing examples of plasticity. Bluehead wrasse, for example, can change gender. Females can become large, colorful, territorial males. Other bluehead wrasse exist as “sneaker” males that look identical to the females. This camouflage strategy enables them to sneak in undetected and spawn with the females within the territories of larger males.
The gender a young bluehead wrasse will manifest depends upon the number of individuals it encounters when it is first developing. If it hatches on a large reef where there are so many fish it is difficult for the big males to defend harems of females, then the developing baby is more likely to become a small sneaker male. “Social interactions really early in life set off these trajectories of whether they become a female, whether they become a male, or whether they become a female that then changes sex to become a big male,” says Munday. “This one species has all these morphologies, all these alternative strategies, all this plasticity—it’s just incredible.”

Bluehead wrasse (Thalassoma bifasciatum) have the inherent ability to become either male or female. Their gender is determined largely by the social dynamics on the reef. Photo by Alex Mustard/Minden Pictures
Munday is quick to point out that just because one sees advantageous plasticity in one trait, it doesn’t mean it will exist in all traits. “The spiny chromis damselfish offspring were perfectly capable of getting enough oxygen in those higher water temperatures,” he explains. “But they did not breed.”
Here is where the hopeful story of damselfish resilience to warming ocean temperatures suddenly veers toward despair. Or does it? Thanks to a smart-thinking postdoctoral fellow in Munday’s lab, the experiment included an additional treatment option. Rather than just putting the fish directly into water that is 3 °C warmer than normal, Jennifer Donelson put fish into water 1.5 °C warmer for the first year and then into water 3 °C warmer the second year.
And, those fish did breed.
A gradual rate of temperature change enabled the damselfish to acclimate in ways they couldn’t do in response to a sudden increase. In other words, given enough time to adjust, the kids were all right.
Whether or not coral reef fish can deal with sudden versus gradual ocean temperature changes is a crucial question given the escalating frequency and intensity of heatwaves associated with climate change. In October 2018, the world’s leading climate scientists—the Intergovernmental Panel on Climate Change—released their most dire report ever: the world is currently 1 °C warmer than preindustrial levels and every fraction of additional warming will worsen the devastating impact of climate instability. Thirty percent of corals in the Great Barrier Reef died in a horrific nine-month marine heatwave in 2016. An increase in just one to two degrees causes corals to bleach. “If they stay bleached, they will die,” Munday explains. “In 2016 and 2017 we had the worst back-to-back bleaching the Great Barrier Reef has ever seen.”
Do corals themselves have the potential to gain resilience from their parents’ experiences? In a series of experiments at Hollie Putnam’s lab at the University of Rhode Island, researchers exposed adult corals to increased temperature and acidification, and then exposed their offspring to the same situation. “We found that there is potential for beneficial acclimatization,” she says. “The offspring show greater survivorship and growth rates if the parents have been previously exposed to short periods of adverse conditions.” Putnam believes that here, too, the rapid adaptation is due to epigenetics.

Although corals are highly susceptible to marine heatwaves, there is some evidence that, at least in the short term, adult coral exposed to higher temperatures can pass some ability to acclimatize to their offspring. Photo by Jurgen Freund/Minden Pictures
But how long will these benefits last? Is the improved capacity to live in warmer water a fleeting phenomenon, like Cinderella’s pumpkin coach, or is it passed on through future generations to come?
Thousands of kilometers north, and on the other side of the globe, scientists in Maritime Canada studying winter skate—the fish not the hockey equipment—found an intriguing way to explore that question. They studied genetic material from two populations of these cartilaginous fish—one that lives off the exposed coast of Nova Scotia in chilly Atlantic waters and one that lives in waters 10 °C warmer in the southern Gulf of St. Lawrence. The winter skate in the St. Lawrence are dramatically smaller, an adaptation that enables them to live in the lower oxygen conditions of the warmer water. Their small size was achieved by epigenetically modifying their genes to make a physiological change.
These two groups have been geographically separated for 7,000 years, yet they are genetically indistinguishable. Technically, the smaller skate have not evolved—their DNA is identical to the other population of skate. Instead, they’ve adapted their bodies to the warmer water by making a total of 3,653 changes in their gene expression patterns. These epigenetic changes have developed and persisted over the course of 318 generations.
And yet, because they do not involve permanent changes to the genetic DNA code, epigenetic changes are also potentially reversible—a quality that is driving a revolution in human medicine. “The great potential for epigenetic therapies,” writes Nita Ahuja and her colleagues from Johns Hopkins University School of Medicine in a 2016 paper in the Annual Review of Medicine “lies in the fact that, unlike genetic abnormalities, epigenetic changes are reversible, allowing recovery of function for affected genes with normal DNA sequences. These therapies aim to reprogram cancer cells to a more normal state.”

Although they are the same species with identical DNA, two populations of winter skate (Raja ocellata) physically separated for thousands of years are morphologically quite different. The population living in warmer waters with low oxygen is physically smaller than that living in colder, oxygen-rich waters. Photo by Fred Bavendam/Minden Pictures
Today a vast collection of medical and biological studies demonstrates how changes in temperature and hormones or other chemicals affect DNA by modifying certain epigenetic factors which instruct the genes whether or not to be expressed. It’s a far cry from what people understood over a century ago about how environments create change. Back in the 1880s, German biologist August Weismann sought to understand how changes to one generation might affect the next by amputating the tails of five successive generations of mice. He concluded that the effects of an environmental stimulus (in this case, cutting off a tail) could not be transmitted to its offspring (the mice were all born with tails).
What Weismann failed to realize is that cutting off a parent’s tail has nothing to do with whether or not a mouse will be born without a tail. Temperature or chemicals, not physical injury, trigger epigenetic factors. Plus, the capacity to be born without tails would have had to already exist within the genetic code of mice in order for this trait to ever be passed along or expressed.
That said, astonishing examples of transgenerational resilience that defy belief continue to be discovered. For example, Mylene Mariette, a behavioral ecologist at Deakin University in Australia, was observing relationships between zebra finch parents when she heard a call she didn’t recognize. Although she did not realize it at the time, it was the sound of a zebra finch calling to its chicks—chicks that hadn’t hatched yet. Zebra finch parents were communicating to their babies still inside the egg.
“At first I didn’t know the call had anything to do with temperature,” Mariette explains. “When you discover something really new, you don’t have a theory to go with it. It was quite an adventure.” Yet after listening to 600 hours of recordings, she determined that zebra finch parents only make this call when it’s super hot outside and they’re panting to cope with the heat. “The parents mostly make the ‘hot calls’ near the end of the incubation period,” she says. “We don’t know why, but perhaps the embryos have not developed the ability to hear before then.”
In the experiments that followed, Mariette and her research colleagues placed eggs in incubators and divided them into two groups. One group heard typical parent-to-parent calls and the other was played recordings of parents making hot calls for the last five days before hatching.
The chicks showed no difference in body size at hatching. Yet within 24 hours of being placed in either a cool or hot nest, the research team could already detect a difference in the chicks’ weights. And it continued throughout their development. Chicks that had heard the hot calls as embryos grew more slowly and had smaller body sizes. Smaller bodies are easier to cool, which is an advantage in hot temperatures. When they grew up, the hot call birds went on to prefer warmer nesting spots. And they had more babies than the birds that hadn’t been primed for the heat.

Experiments have shown that the size of zebra finch (Taeniopygia guttata) chicks and their ability to withstand warmer temperatures is determined by the parents’ exposure to heat. Photo by Martin Willis/Minden Pictures
In short, the research indicates that chicks that hear hot calls during the time their temperature regulation system is forming develop differently. It’s the first time such profound effects of prenatal acoustic experience have been documented. Zebra finches appear to be using particular songs that alter the unhatched chicks’ temperature regulation system and change their sensitivity to heat over the long term. They seem to prepare their unhatched chicks to deal with high temperature extremes—the kinds of temperatures we expect from climate change.
“My research makes me hopeful,” Mariette says, “because it really shows that birds are better at dealing with changes to their environment than we think. Perhaps they will be able to deal with increasing temperatures and all those other environmental changes that are happening too quickly.”
I am awed by the capacity of damselfish and corals and zebra finch parents to positively impact the resilience of their offspring in these daunting circumstances. Such unexpected abilities make me hungry to better understand the mechanisms of intergenerational resilience in other species, and across human circumstances.
Recognizing transgenerational resilience is inextricably linked with hope. It offers possibility, even in the midst of dire circumstances. The other day, while speaking with my friend Lillian Howard, a strong advocate for Indigenous, social, and environmental justice, I tell her how much it matters to me to know that other species gain resilience from the experiences of their parents. It fuels my already fervent passion to reduce carbon dioxide emissions. She responds with a warm smile. “Everything is interconnected,” Howard reminds me. “Our ancestors are always with us.”

